 
The most notable realized oxidation state is +8 in ruthenium, xenon, osmium, iridium, hassium, and some complexes including plutonium tetroxides the least realized oxidation state is 4 for certain carbon group elements. 
The normal oxidation state of an element can play a role at times. Whole numbers are frequently used to refer to oxidation states, which can be positive, negative, or zero. The level of oxidation for an atom in a chemical compound is shown by its oxidation state it is the speculative charge that an atom would have if all bonds to atoms of various elements were completely ionic. Oxidation state and trends in chemical reactivity Because of their low reactivity, these metals do not dissolve easily in strong acids. Platinum has the lowest reactivity, followed by gold. Noble metals are metals with low reactivity. Metals can exist in multiple oxidation states and have intermediate reactivity. Helium is the least reactive element in this group, forming no stable compounds. Noble gases are the least reactive elements. Cesium, which is directly above francium on the periodic table, is the most reactive metal with a stable isotope. However, francium is a radioactive element that is only found in trace amounts. The most reactive metal is francium, which is also the last alkali metal (and the most expensive element). The halogen group’s first element, fluorine, is the most reactive. Alkali metals, halogens, and alkaline earth metals all have high reactivity. These elements are found in the periodic table’s top right and lower left corners, as well as in certain element families. 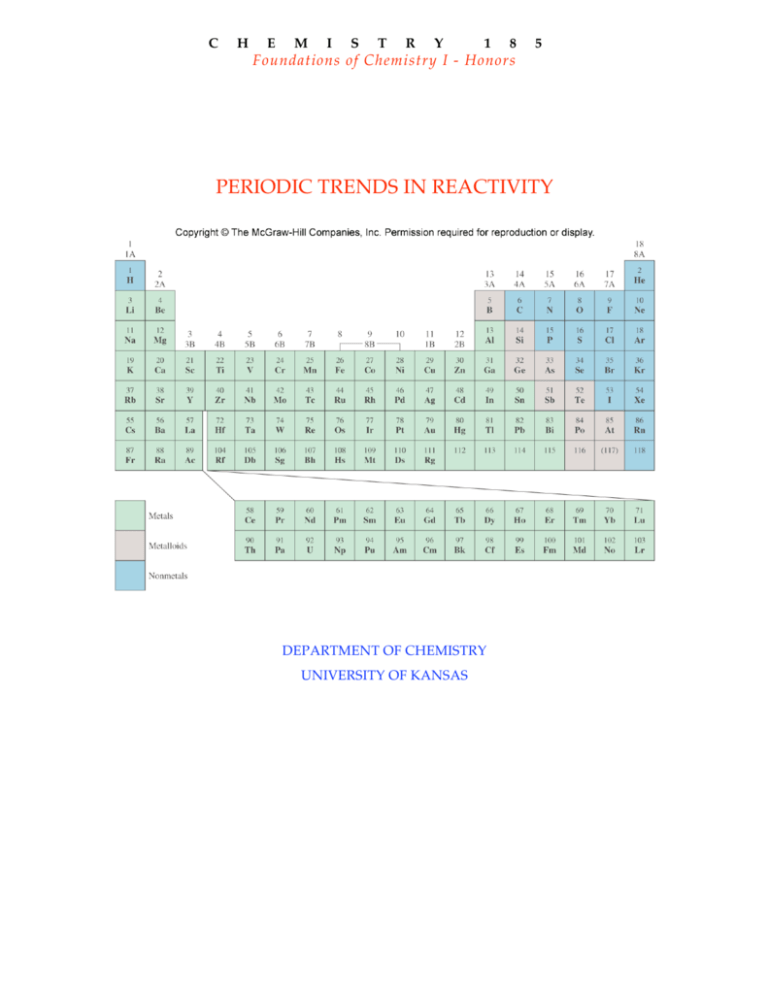
Highly electropositive and highly electronegative materials have a great potential to react with one another. The periodic table’s arrangement of elements allows reactivity predictions to be made. 
Reactivity can also be defined as the scientific study of chemical reactions and their kinetics. Temperature increases the amount of energy available for a chemical reaction, making it more likely. The temperature has an effect on reactivity. They typically burn in both water and oxygen in the air. The elements and compounds with the highest reactivity have the ability to spontaneously or explosively ignite. The reaction can involve the substance alone or in combination with other atoms or compounds, and it is usually accompanied by the release of energy. Reactivity is a measure of how easily a substance undergoes a chemical reaction in chemistry. The Periodic Law also influenced the creation of the periodic table. The Periodic Law’s discovery was one of the most momentous events in the history of chemical science. The periodic recurrence of elements with comparable physical and chemical properties derives directly from the periodic recurrence of identical electronic configurations in respective atoms’ outer shells when elements are enumerated in increasing atomic number order. The Periodic Law had no scientific explanation at first, and it was solely utilized as an empirical principle but, with the development of quantum physics, the theoretical basis for the Periodic Law could be understood. Lothar Meyer’s table was presented several months after Mendeleev’s, but he disagreed with Mendeleev’s Periodic law. In 1913, Henry Moseley found that the atomic number, not the atomic weight, determines periodicity. Mendeleev also proposed an elemental periodic system based on the chemical and physical properties of the elements and their compounds, rather than only atomic weights. After a number of nineteenth-century scientific investigations, Russian chemist Dmitri Mendeleev discovered this principle in 1871. Many of the physical and chemical properties of lithium, such as its vigorous reactivity with water, recur in sodium, potassium, and cesium after arranging elements in increasing atomic numbers. The Periodic Law asserts that when chemical elements are enumerated in increasing atomic number order, many of their properties undergo cyclical changes, with comparable elements reoccurring at regular intervals. According to this criterion, pyridine is more aromatic than furan, but it is difficult to say by how much. As a result, the degree of aromaticity is measured in terms of reactivity by the relative tendency toward substitution rather than addition. The extra stability, in turn, determines the compound’s tendency to react by substitution of hydrogen-that is, replacement of a singly bonded hydrogen atom with another singly bonded atom or group-rather than the addition of one or more atoms to the molecule via the breaking of a double bond (see substitution reaction addition reaction). 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
